
The density of sodium chloride at 25^(@)C "is" 2.163 xx 10 ^(3) " kg m" ^(-3) When X -rays rom a palladium target having waveleth of 58.1 pm are used, the (200)
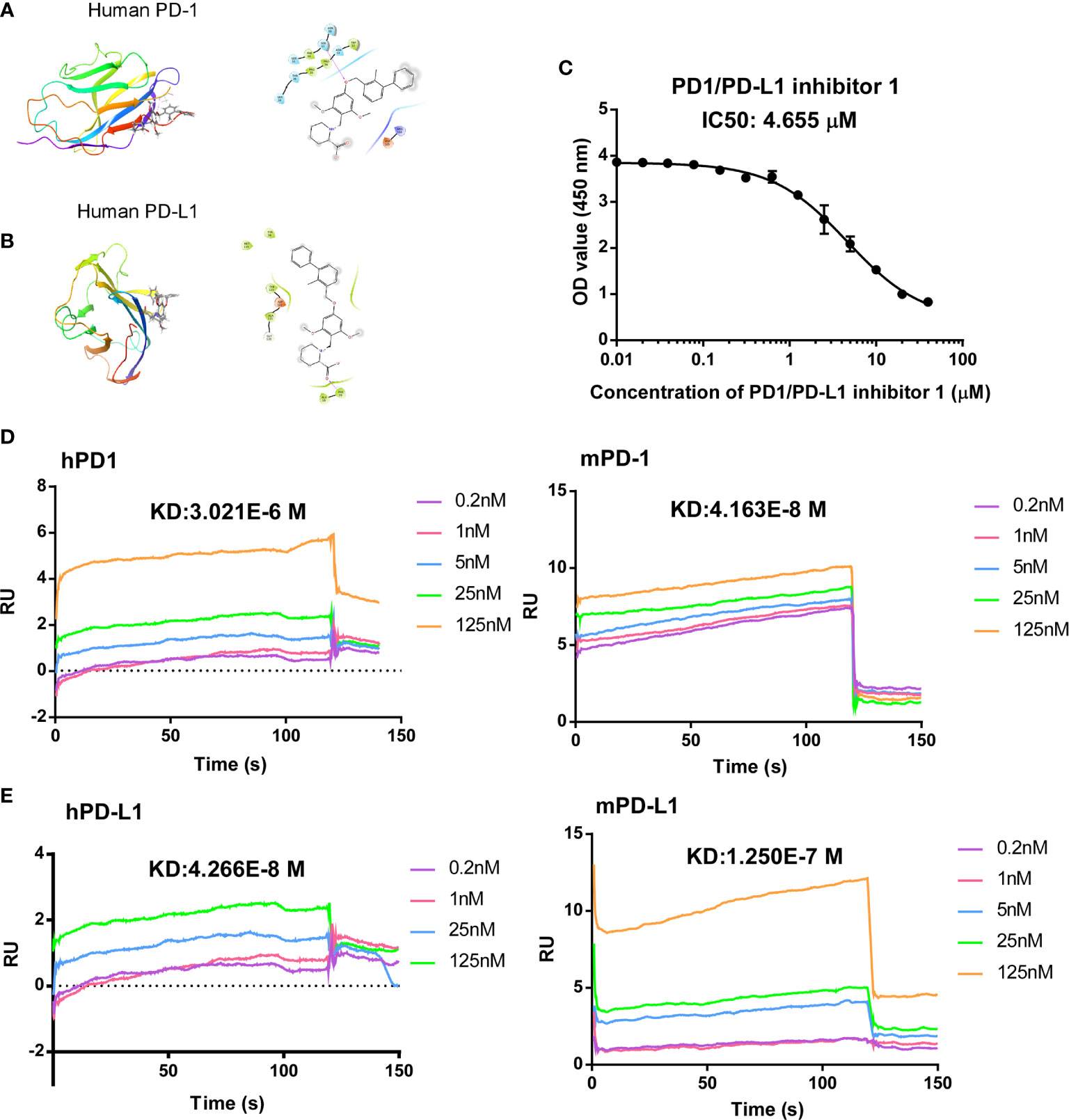
Frontiers | A Small Molecule Antagonist of PD-1/PD-L1 Interactions Acts as an Immune Checkpoint Inhibitor for NSCLC and Melanoma Immunotherapy

Average phonon frequency (hxi) vs. density (q) for palladium in a... | Download High-Quality Scientific Diagram

Synthesis and Characterization of Phosphinobenzenamine Palladium Complexes and Their Application in Ethylene Polymerization and Copolymerization with Polar Monomers | Organometallics

SOLVED: A metal crystallizes in the face-centered cubic (FCC) lattice. The density of the metal is 8902 kglm , and the length of a unit cell edge, a,is 352.4 pm. Calculate the













:max_bytes(150000):strip_icc()/palladium-56a129313df78cf77267f7e3.jpg)




![PDF] Palladium-Based Catalysts for Ethanol Electrooxidation in Alkaline Media | Semantic Scholar PDF] Palladium-Based Catalysts for Ethanol Electrooxidation in Alkaline Media | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2fa46485c976bbb79df9889b10d273b6a13648f2/21-Table1-1-1.png)