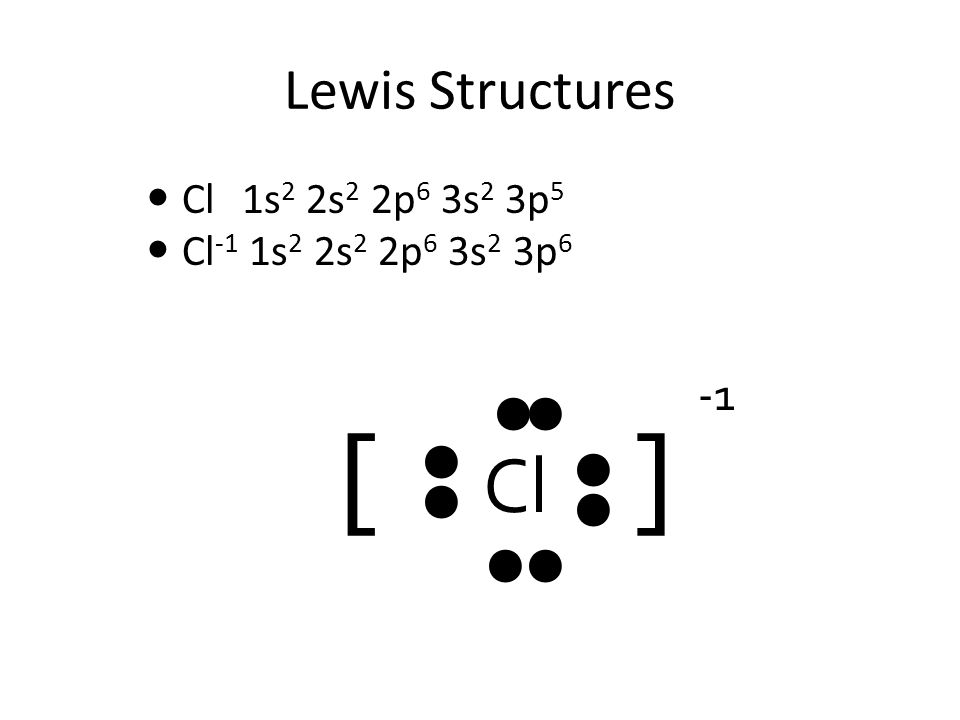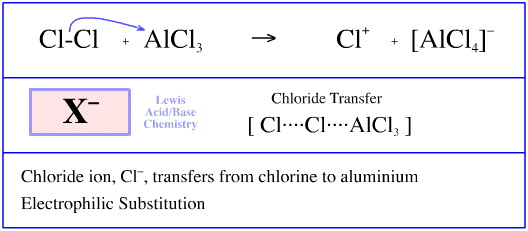
Is the following acid-base reaction Arrhenius, Bronsted-Lowry, or Lewis: AlCl3 + Cl --> AlCl4- | Socratic
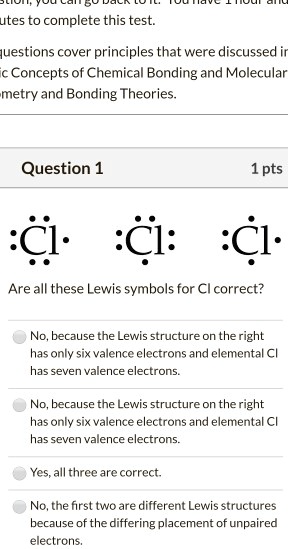
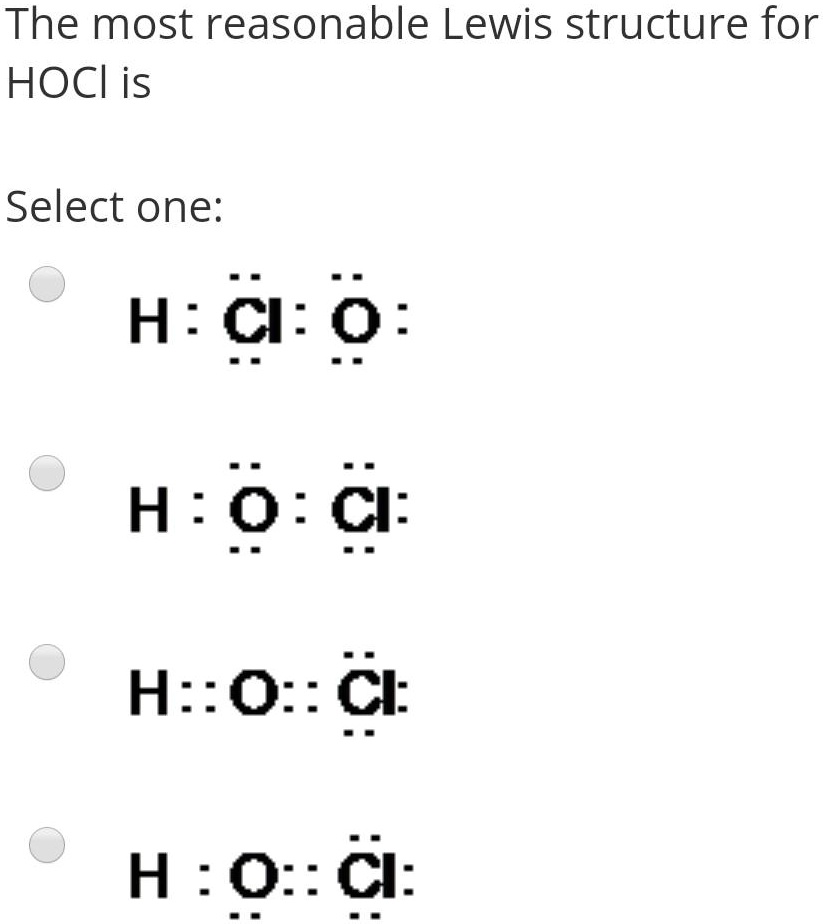
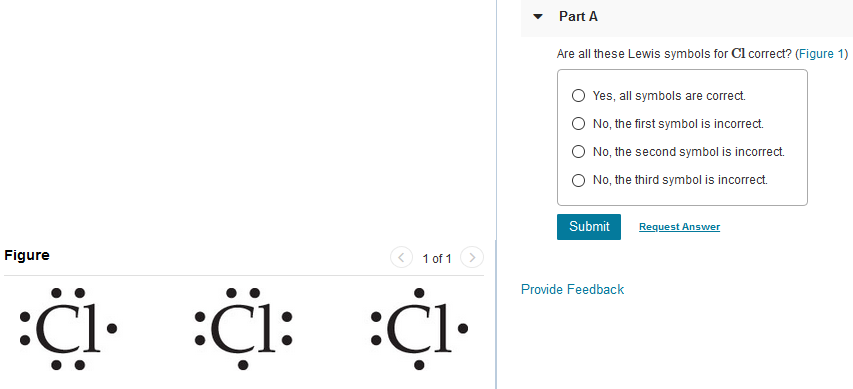
SOLVED:utes to complete this test: juestions cover principles that were discussed ir Concepts of Chemical Bonding and Molecular metry and Bonding Theories: Question 1 1pts Cl :Cl: 'Cl: Are all these Lewis