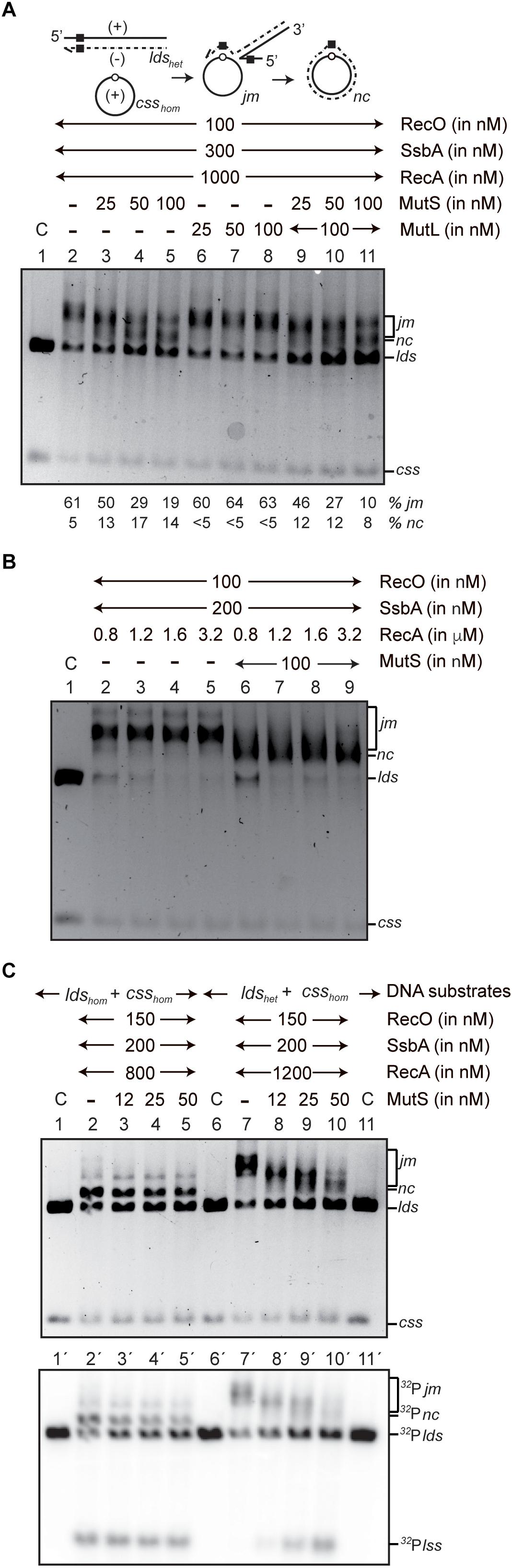
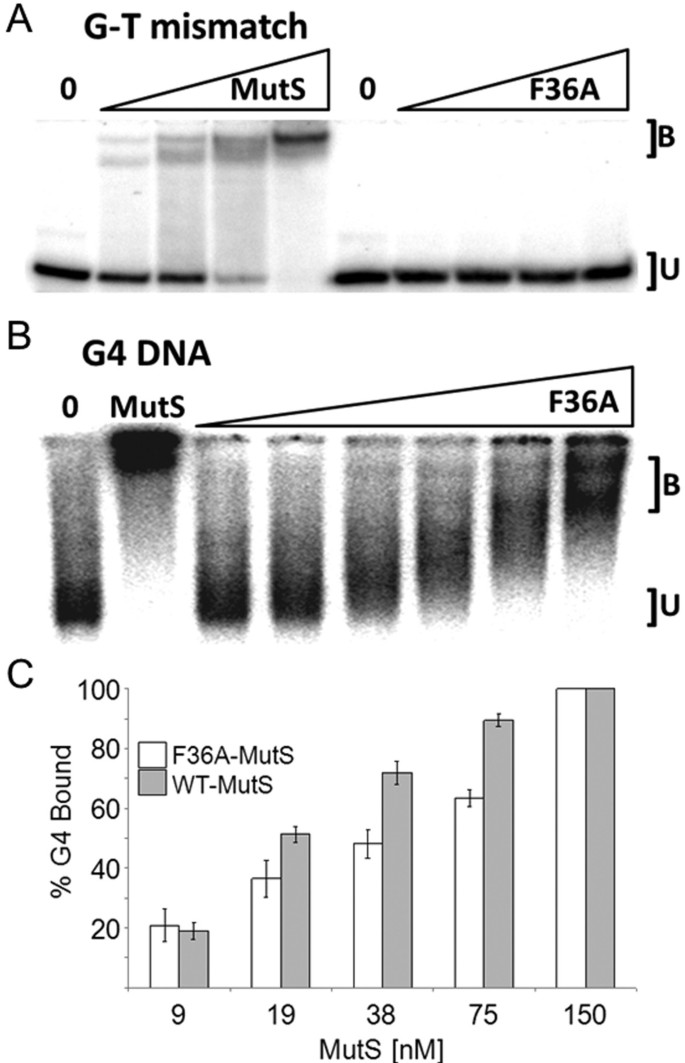
Role of MutS ATPase Activity in MutS,L-dependent Block of in Vitro Strand Transfer* - Journal of Biological Chemistry
PLOS ONE: Epistatic Roles for Pseudomonas aeruginosa MutS and DinB (DNA Pol IV) in Coping with Reactive Oxygen Species-Induced DNA Damage

The effects of nucleotides on MutS-DNA binding kinetics clarify the role of MutS ATPase activity in mismatch repair. | Semantic Scholar

Polypyrimidine Tract Binding Protein Prevents Activity of an Intronic Regulatory Element That Promotes Usage of a Composite 3′-Terminal Exon - ScienceDirect
Evolution of Pseudomonas aeruginosa toward higher fitness under standard laboratory conditions | The ISME Journal
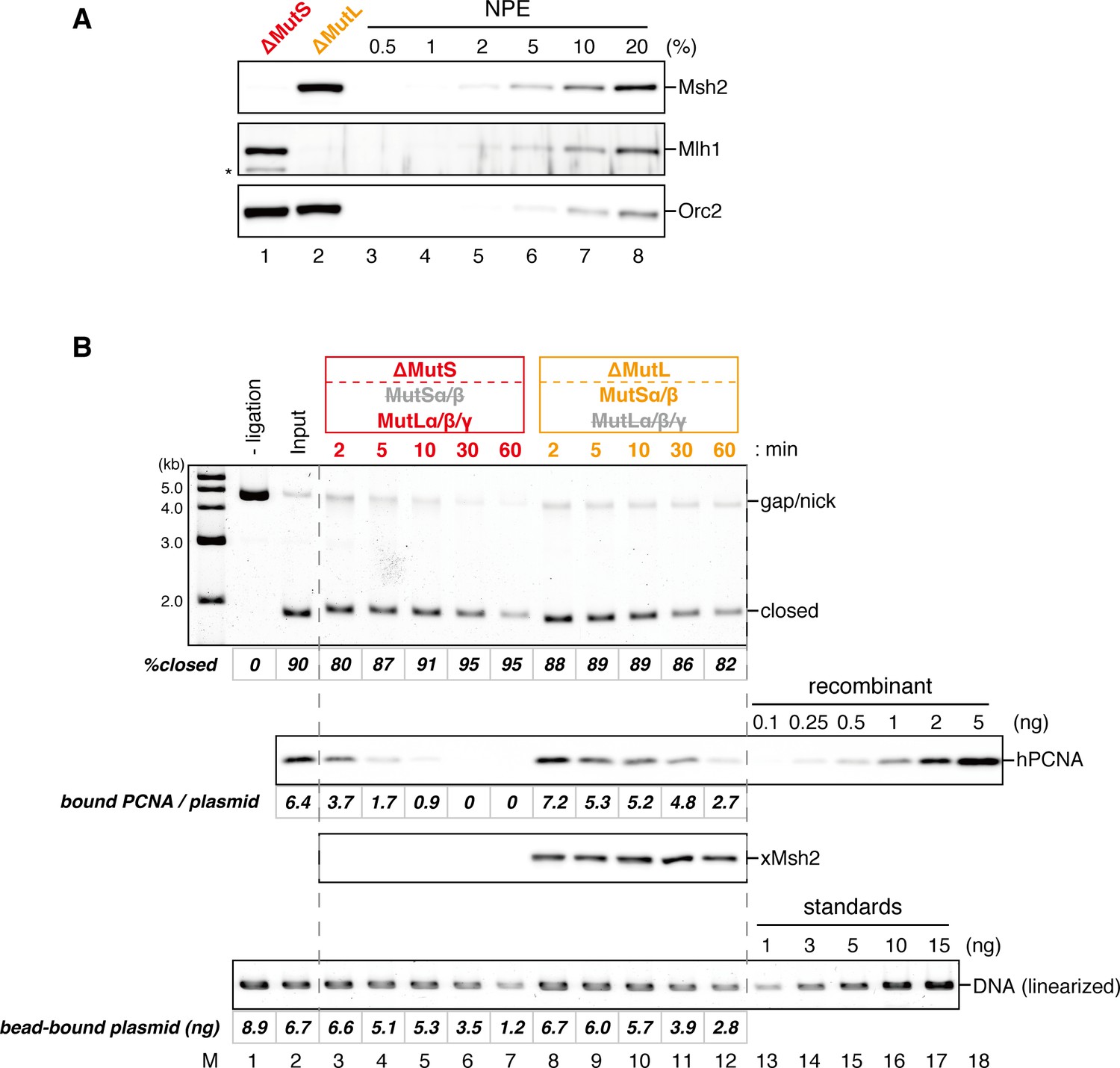
Figures and data in MutSα maintains the mismatch repair capability by inhibiting PCNA unloading | eLife

Figures and data in MutSα maintains the mismatch repair capability by inhibiting PCNA unloading | eLife

Binding of MutS and MutS to ATP or ADP. MutS (A, C, and E) or MutS (B,... | Download Scientific Diagram

New insights into how MutS separates its function in the regulation of the Pol IV access to replication sites from that in the conserved MMR pathway | bioRxiv

Emergence of the Novel Aminoglycoside Acetyltransferase Variant aac(6′)-Ib-D179Y and Acquisition of Colistin Heteroresistance in Carbapenem-Resistant Klebsiella pneumoniae Due to a Disrupting Mutation in the DNA Repair Enzyme MutS | mBio